Where Our Optics Are Used
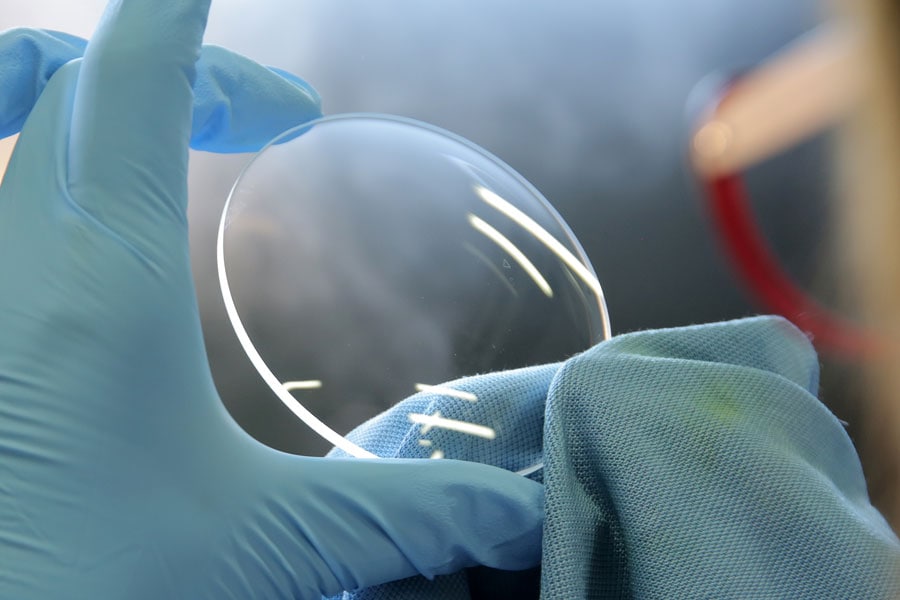
Sterling Precision Optics is a optical manufacturing company serving a diverse array of industries, including aerospace, medical, defense, analytical instruments, detectors, machine vision, underwater photography, and more.
Our optical components are integral to various equipment utilized in aerospace, medical, defense, and scientific fields. For instance, in the medical sector, our custom optical lenses are vital components of medical scopes, and we engineer custom optical filter glass that enhances vein visibility for medical professionals. Additionally, we produce optical filters and custom lenses for blood gas analyzers and other analytical instruments, along with UV filters essential for UV curing systems in the dental industry.
In aerospace, our contributions include crafting gauge covers, anti-reflective covers for instrument panels, and tempered port windows for aircraft taxi lights. Our lenses are also employed in high-altitude cameras onboard aircraft. Furthermore, we fabricate fluid level sight glasses for monitoring fluid levels in aerospace applications.
Within defense applications, we specialize in manufacturing night vision filters for goggles and other equipment, as well as IR windows for fire suppression systems. Moreover, our portfolio encompasses optical lenses, windows, mirrors (both hot and cold), and prism assemblies tailored for fire control systems, M1 tank periscopes, and night vision imaging systems (NVIS).
We provide specialized optical solutions tailored to unique requirements for the scientific industry as well. Our capabilities extend beyond standard components to include solutions like precision lenses optimized for specific medical scopes, advanced optical filters for improved vein detection, and high-performance lenses and optical assemblies for aerospace high-altitude imaging systems. We also collaborate closely with researchers and institutions to deliver specialized optical components for metrology, spectroscopy, and imaging applications, facilitating precise measurement and analysis across diverse disciplines.
Sterling is a full service custom optical and glass company
Custom Products
Select a product type for more
Why Work With Us?
We have been proudly serving the optical community since 1958. We are quick with our response time for R.F.Qs. We are on time with our deliveries. We are quality conscious to your specifications and hold ourselves to our ISO 9001:2015 certification....
 ISO 9001:2015 Certified
ISO 9001:2015 Certified
 Fabrication and Testing
Fabrication and Testing
 Equipment
Equipment


 CUSTOM OPTICAL FILTERS
CUSTOM OPTICAL FILTERS
 OPTICAL WINDOWS
OPTICAL WINDOWS
 OPTICAL COATINGS
OPTICAL COATINGS
 UV OPTICS
UV OPTICS
 CYLINDRICAL OPTICS
CYLINDRICAL OPTICS
 CUSTOM TEMPERED OPTICS
CUSTOM TEMPERED OPTICS
 OPTICAL MIRRORS
OPTICAL MIRRORS
 NEUTRAL DENSITY
NEUTRAL DENSITY
 PRISMS & RETROREFLECTORS
PRISMS & RETROREFLECTORS
 ASSEMBLIES
ASSEMBLIES
 OPTICAL LENSES
OPTICAL LENSES
 NIGHT VISION FILTERS
NIGHT VISION FILTERS
 ACHROMATIC LENSES
ACHROMATIC LENSES
 OPTICAL BEAM SPLITTERS
OPTICAL BEAM SPLITTERS
